4.1 Heat & Energy
What is Internal Energy?
All matter (solids, liquids, and gases) is made of particles (atoms and molecules). Internal Energy is the total energy stored inside a system.
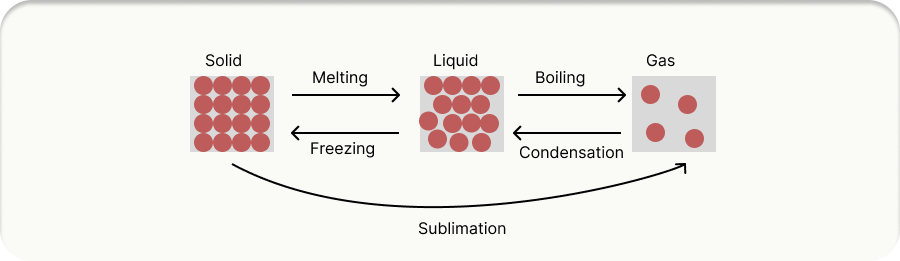
All matter (solids, liquids, and gases) is made of particles (atoms and molecules). Internal Energy is the total energy stored inside a system.
⚡ Internal Energy =
Kinetic Energy of particles (from their movement)
+
Potential Energy of particles (from their position relative to each other)
+
Potential Energy of particles (from their position relative to each other)
Solid
Particles vibrate in fixed positions. Strong bonds. Lowest energy.
Liquid
Particles move around each other. Weaker bonds. More energy.
Gas
Particles move freely and fast. No bonds. Highest energy.
💡 Key Concept: Heating an Object
Changes of State:
Heating an object increases its internal energy. This can either:
1. Increase temperature → particles move faster (↑ kinetic energy)
2. Change state → particles break bonds (↑ potential energy)
During a change of state, temperature stays constant.
1. Increase temperature → particles move faster (↑ kinetic energy)
2. Change state → particles break bonds (↑ potential energy)
During a change of state, temperature stays constant.

Why Temperature Stays Constant During State Changes:
When ice melts at 0°C:
• Energy is being added
• But temperature stays at 0°C
• Energy is used to break bonds between particles
• This increases potential energy, not kinetic energy
• Once all bonds are broken, temperature rises again
• Energy is being added
• But temperature stays at 0°C
• Energy is used to break bonds between particles
• This increases potential energy, not kinetic energy
• Once all bonds are broken, temperature rises again
🎯 States of Matter Quiz
What is Specific Heat Capacity?
The Specific Heat Capacity (c) of a substance is the amount of energy needed to raise the temperature of 1 kg of that substance by 1°C.
The Specific Heat Capacity (c) of a substance is the amount of energy needed to raise the temperature of 1 kg of that substance by 1°C.
⚡ Specific Heat Capacity Formula:
$$\Delta E = m \times c \times \Delta\theta$$
Where:
• $\Delta E$ = energy transferred (J)
• $m$ = mass (kg)
• $c$ = specific heat capacity (J/kg°C)
• $\Delta\theta$ = change in temperature (°C)
Where:
• $\Delta E$ = energy transferred (J)
• $m$ = mass (kg)
• $c$ = specific heat capacity (J/kg°C)
• $\Delta\theta$ = change in temperature (°C)
Water
4,200
J/kg°C
High - heats slowlyAluminium
900
J/kg°C
Low - heats quicklyCopper
385
J/kg°C
Very low - heats very quickly
💡 What Does This Mean?
• High SHC (like water): Takes a lot of energy to heat up, also takes a long time to cool down
• Low SHC (like metals): Heats up very quickly, also cools down quickly
This is why water is used in heating systems - it stores lots of energy.
• Low SHC (like metals): Heats up very quickly, also cools down quickly
This is why water is used in heating systems - it stores lots of energy.
Example 1: Heating Water
How much energy is needed to heat 2 kg of water from 20°C to 100°C?
(Specific heat capacity of water = 4,200 J/kg°C)
Step 1: Find the temperature change
$\Delta\theta = 100 - 20 = 80°C$
Step 2: Write the formula
$\Delta E = m \times c \times \Delta\theta$
Step 3: Substitute values
$\Delta E = 2 \times 4200 \times 80$
Step 4: Calculate
$\Delta E = 672,000$ J = 672 kJ
(Specific heat capacity of water = 4,200 J/kg°C)
Step 1: Find the temperature change
$\Delta\theta = 100 - 20 = 80°C$
Step 2: Write the formula
$\Delta E = m \times c \times \Delta\theta$
Step 3: Substitute values
$\Delta E = 2 \times 4200 \times 80$
Step 4: Calculate
$\Delta E = 672,000$ J = 672 kJ
Example 2: Finding Temperature Change
A 0.5 kg aluminium block absorbs 9,000 J of energy. What is the temperature rise?
(Specific heat capacity of aluminium = 900 J/kg°C)
Step 1: Rearrange the formula
$\Delta\theta = \frac{\Delta E}{m \times c}$
Step 2: Substitute values
$\Delta\theta = \frac{9000}{0.5 \times 900}$
Step 3: Calculate
$\Delta\theta = \frac{9000}{450} = 20°C$
(Specific heat capacity of aluminium = 900 J/kg°C)
Step 1: Rearrange the formula
$\Delta\theta = \frac{\Delta E}{m \times c}$
Step 2: Substitute values
$\Delta\theta = \frac{9000}{0.5 \times 900}$
Step 3: Calculate
$\Delta\theta = \frac{9000}{450} = 20°C$
🧮 Specific Heat Capacity Calculator:
Mass: kg
SHC: J/kg°C
ΔT: °C
🎯 Specific Heat Capacity Practice
What is Specific Latent Heat?
Specific Latent Heat (L) is the amount of energy needed to change the state of 1 kg of a substance without any change in temperature.
• Melting (solid → liquid)
• Freezing (liquid → solid)
Water: 334,000 J/kg
• Boiling (liquid → gas)
• Condensing (gas → liquid)
Water: 2,260,000 J/kg
Specific Latent Heat (L) is the amount of energy needed to change the state of 1 kg of a substance without any change in temperature.
⚡ Specific Latent Heat Formula:
$$E = m \times L$$
Where:
• $E$ = energy transferred (J)
• $m$ = mass (kg)
• $L$ = specific latent heat (J/kg)
Where:
• $E$ = energy transferred (J)
• $m$ = mass (kg)
• $L$ = specific latent heat (J/kg)
Latent Heat of Fusion
Energy to change between solid ↔ liquid• Melting (solid → liquid)
• Freezing (liquid → solid)
Water: 334,000 J/kg
Latent Heat of Vaporisation
Energy to change between liquid ↔ gas• Boiling (liquid → gas)
• Condensing (gas → liquid)
Water: 2,260,000 J/kg
💡 Why "Latent" Heat?
"Latent" means "hidden". The energy is hidden because:
• Temperature doesn't change during state change
• Energy goes into breaking/forming bonds
• A thermometer won't detect this energy transfer
Note: Vaporisation requires much more energy than fusion because gas particles must completely overcome all attractive forces.
• Temperature doesn't change during state change
• Energy goes into breaking/forming bonds
• A thermometer won't detect this energy transfer
Note: Vaporisation requires much more energy than fusion because gas particles must completely overcome all attractive forces.
Example 1: Melting Ice
How much energy is needed to melt 0.5 kg of ice at 0°C?
(Specific latent heat of fusion for water = 334,000 J/kg)
Step 1: Write the formula
$E = m \times L$
Step 2: Substitute values
$E = 0.5 \times 334,000$
Step 3: Calculate
$E = 167,000$ J = 167 kJ
(Specific latent heat of fusion for water = 334,000 J/kg)
Step 1: Write the formula
$E = m \times L$
Step 2: Substitute values
$E = 0.5 \times 334,000$
Step 3: Calculate
$E = 167,000$ J = 167 kJ
Example 2: Boiling Water
How much energy is needed to turn 2 kg of water at 100°C into steam?
(Specific latent heat of vaporisation = 2,260,000 J/kg)
Step 1: Write the formula
$E = m \times L$
Step 2: Substitute values
$E = 2 \times 2,260,000$
Step 3: Calculate
$E = 4,520,000$ J = 4,520 kJ or 4.52 MJ
(Specific latent heat of vaporisation = 2,260,000 J/kg)
Step 1: Write the formula
$E = m \times L$
Step 2: Substitute values
$E = 2 \times 2,260,000$
Step 3: Calculate
$E = 4,520,000$ J = 4,520 kJ or 4.52 MJ
Example 3: Combined Problem
How much energy is needed to heat 0.2 kg of ice from -10°C to water at 50°C?
(c for ice = 2,100 J/kg°C, c for water = 4,200 J/kg°C, L fusion = 334,000 J/kg)
Step 1: Heat ice from -10°C to 0°C
$E_1 = 0.2 \times 2100 \times 10 = 4,200$ J
Step 2: Melt ice at 0°C
$E_2 = 0.2 \times 334,000 = 66,800$ J
Step 3: Heat water from 0°C to 50°C
$E_3 = 0.2 \times 4200 \times 50 = 42,000$ J
Total: $E = 4,200 + 66,800 + 42,000 = 113,000$ J = 113 kJ
(c for ice = 2,100 J/kg°C, c for water = 4,200 J/kg°C, L fusion = 334,000 J/kg)
Step 1: Heat ice from -10°C to 0°C
$E_1 = 0.2 \times 2100 \times 10 = 4,200$ J
Step 2: Melt ice at 0°C
$E_2 = 0.2 \times 334,000 = 66,800$ J
Step 3: Heat water from 0°C to 50°C
$E_3 = 0.2 \times 4200 \times 50 = 42,000$ J
Total: $E = 4,200 + 66,800 + 42,000 = 113,000$ J = 113 kJ
| Substance | Latent Heat of Fusion (J/kg) | Latent Heat of Vaporisation (J/kg) |
|---|---|---|
| Water | 334,000 | 2,260,000 |
| Ethanol | 108,000 | 855,000 |
| Lead | 23,000 | 871,000 |
🧮 Latent Heat Calculator:
Mass:
kg
Latent Heat:
J/kg
🎯 Latent Heat Practice
Real Life Applications:
• Sweating: Evaporation removes heat from skin (uses latent heat of vaporisation)
• Ice packs: Melting ice absorbs heat from injuries
• Steam burns: Worse than water burns because steam releases latent heat when condensing
• Refrigerators: Use evaporation and condensation to move heat
• Weather: Huge amounts of energy stored in water vapour in clouds
• Sweating: Evaporation removes heat from skin (uses latent heat of vaporisation)
• Ice packs: Melting ice absorbs heat from injuries
• Steam burns: Worse than water burns because steam releases latent heat when condensing
• Refrigerators: Use evaporation and condensation to move heat
• Weather: Huge amounts of energy stored in water vapour in clouds